According to the National Stroke Association, full recovery occurs in only 10 percent of people who have a stroke, with 25 percent recovering with minor impairments. Approximately 40 percent experience moderate to severe physical and cognitive issues that end up requiring special care. And 10 percent of stroke patients end up in long-term care in a health facility or nursing home.
Traditionally, stroke rehabilitation in hospitals starts quickly, usually within 24 to 48 hours. And rehab can be a very frustrating experience for the patient. “If they’re in for their first stroke, we want to constantly monitor their vitals because sometimes physical therapy can be very frustrating and it can cause their blood pressure to go up,” says Josephine Lee, an occupational therapist at Memorial Care Long Beach Medical Center in Long Beach, CA.
Therapeutic exercises focus on range of motion, balance, motor control, strength and endurance. If the patient has suffered partial paralysis of an arm or leg (or both) and/or experiences lack of sensation in those limbs, the therapist works intensively to help them regain some control of the affected limb.
In the past, although rehabilitating the affected limb(s) was important, there was a tendency to focus more heavily on “compensatory training” – helping the patient learn to use the unaffected hand to accomplish daily tasks such as getting dressed, brushing teeth and combing hair – something that would get them more quickly functional at home.
But all that changed with the introduction of more effective rehabilitation therapies, and particularly one method known as constraint-induced movement therapy (CIMT).
The story of CIMT starts back in 1895. Sir Charles Sherrington, an English neurophysiologist, pathologist and Nobel laureate, came to two conclusions: first, spinal reflexes (simple behaviors produced by central nervous system pathways in the spinal cord) are the basic building blocks of voluntary movement, and second, anything affecting spinal reflexes, such as loss of sensation, eliminates the capacity for voluntary movement.
Sherrington came to these conclusions based on experiments he conducted on rhesus monkeys. He found that if all the sensation traveling from the forelimb to the spinal cord and brain was blocked by damage to sensory nerves, even though the monkeys still had intact motor nerves to carry information from the brain and spinal cord back to the muscles, the monkeys didn’t – seemingly couldn’t – use the limb.
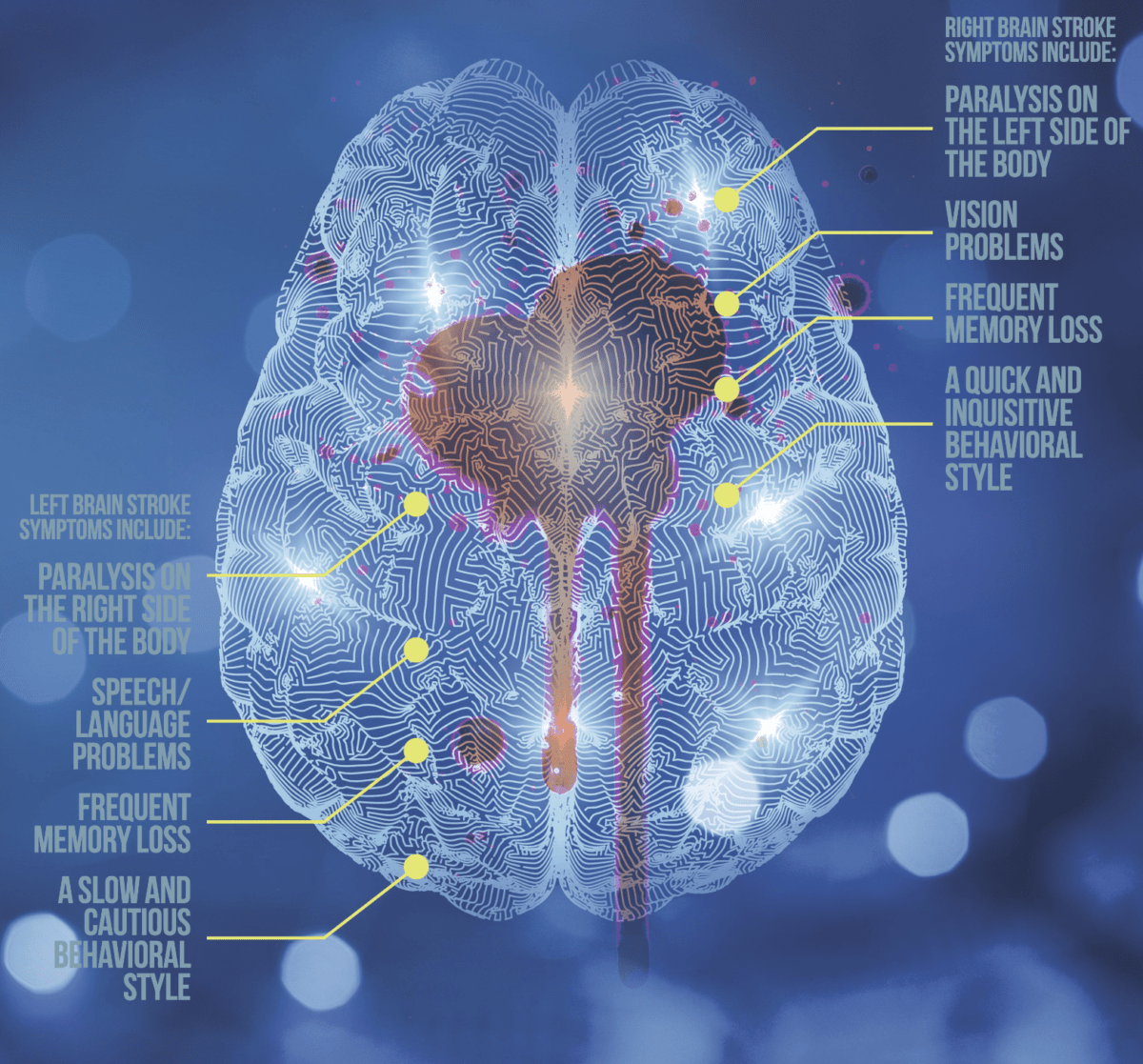
Types of stroke and their effects
Ischemic strokes happen when arterial blood flow to the brain becomes blocked by a blood clot. About 87 percent of all strokes are ischemic. Transient ischemic attack or TIA, often called a “mini stroke,” is caused by a temporary blood clot. TIAs are considered warning strokes, indicating that immediate lifestyle changes are necessary to avoid further issues.
A hemorrhagic stroke occurs when an artery in or surrounding the brain leaks blood or ruptures. The pressure from the leaked blood damages the brain cells. Common causes of hemorrhagic stroke are high blood pressure and aneurysms – bulges in an artery that can stretch and burst.
There are two types of hemorrhagic stroke: intracerebral hemorrhage, in which there is bleeding within the brain, and subarachnoid hemorrhage, a life-threatening type of stroke caused by bleeding into the space surrounding the brain.
The amount of damage a stroke causes depends on the location of the obstruction, the extent of the bleeding and how much brain tissue is affected – which is why getting immediate treatment is so important to long-term recovery.
The central nervous system is wired in a criss-cross or “contra-lateral” fashion, which means the left side of the brain controls the right side of the body and the right side of the brain controls the left side of the body. If a stroke occurs in the left side of the brain, the right side of the body will be affected. If the stroke occurs in the right side of the brain, the left side of the body will be affected. If the stroke occurs toward the back of the brain, the visual centers will most likely be affected.
Paralysis on one side of the body is called hemiplegia; weakness on one side is called hemiparesis.
Changing the paradigm
Up until the 1980s and into the 1990s, it was the dominant view in neurology, neurophysiology and rehabilitation that any interference with the sensory nerves of spinal reflexes eliminated voluntary movement, and that included the case of human patients who had suffered from stroke. Not only was rehabilitation difficult, it was highly unlikely that significant improvement – let alone full recovery – was even possible.
Then, in the early 1990s, along came psychologist and behavioral neuroscientist Dr Edward Taub. “I decided to redo Sherrington’s experiment using behavioral techniques to see if we could force the monkey to use a single deafferented [sensory nerve-depleted] limb,” says Taub. “And that’s the way it turned out. We used conditioned response techniques [training to stimulate a new behavior] and just retrained the forelimb.
“That, by itself, led to an appearance of voluntary movement in the limb. We also used a behavioral technique called shaping.” Shaping, or behavior-shaping, is a behavior modification technique in which gradual, successive approximations to the desired behavior are rewarded.
He also encouraged the monkey to use the affected forelimb by binding the unaffected limb, making it unusable. “Turns out the monkey’s affected limb was clumsy,” he says, “but there was virtually nothing that the monkey couldn’t do.”
Now that he had proven Sherrington’s hypothesis to be incorrect, the next part was figuring out why the monkey didn’t use the affected limb even though it was still completely functional.
“We came up with the concept of learned nonuse to substitute Sherrington’s reflex hypothesis,” he says. This new theory recognized that there is a period of time after the monkey’s limb is desensitized when the limb can’t be used because of something called spinal shock. During that time, the monkey learns to accomplish everything it needs to do with its unaffected limb. By the time the shock has passed and the desensitized limb can actually be used again, the monkey doesn’t use it because it has learned not to – hence the term “learned nonuse.”
Suspecting that the same approach would work with stroke patients suffering from reduced sensory function and partial paralysis, Taub founded the Taub Therapy Clinic and began clinical research at the University of Alabama at Birmingham, working with stroke patients who had suffered partial paralysis in their upper body extremities. Taub jokes about the strange language of rehabilitation. “You no longer have a leg or an arm,” he says, “you have a lower extremity or an upper extremity.”
He and his colleagues worked with patients who had “mild to moderate deficits,” which is actually a much graver condition than it sounds because it means that the average patient has only 10 percent of the use of the affected limb in life situations compared to what they had before the stroke.
Using the same basic techniques he’d used with the monkeys, Taub trained his patients to perform tasks such as brushing their teeth with the affected arm, eating with a fork, putting on shoes and socks and combing their hair.
To ensure forced use of the affected limb, patients were sometimes asked to use constraining items on their unaffected hand or arm such as an ICU mitt (used to prevent patients in the ICU from pulling out their IVs) or a sling that holds the unaffected arm close to the body. Hence, the rehabilitation technique was dubbed constraint-induced movement therapy or CIMT, now sometimes shortened to CI therapy.
The constraints used in CI therapy have been modified over the years to ensure they provide maximal stability and physical safety to the patient. Especially effective are the shaping exercises that CI therapy uses. As a behavioral scientist, Taub understood the power of positive reinforcement – especially for stroke victims who are highly prone to depression over the fact that they can’t simply get their previously functional limbs to work correctly.
Even the smallest improvement in doing an exercise – getting the spoon a half-inch closer to the mouth or being able to reach just the slightest bit more in the general direction of the button that needs buttoning – is noted and praised, thereby encouraging the patient to try even harder next time.
The results with these kinds of therapies, says Taub, were “astounding.” On average, using CI therapy, stroke patients recovered up to 52 percent of their original function – more than a five-fold improvement – in just two weeks. This represents a massive improvement over other treatment approaches in the rehab world.
How the brain changes
In 1948, Polish neuroscientist Jerzy Konorski discovered that the neural networks in the brain (interconnecting networks of cells called neurons) could actually change in shape and size. However, it wasn’t until the 1990s that the term “neuroplasticity” came into common usage to describe the brain’s ability to change and regenerate.
In 1998, Bruce S. McEwen, PhD, began to study the impact of stress on the hippocampus of the brain – a part of the brain associated with learning, emotions and particularly the formation of new memories. Not only did he discover that stress can cause neural networks in the brain to shrink, he also discovered that the damage is recoverable through a process called “axonal sprouting.”
In this phenomenon, undamaged axons – the long threadlike part of a neuron that conducts electrical impulses from one cell to another – grow new nerve endings to replace those that were injured or severed.
Advances in medical imaging techniques to assess brain structure and function have shown that neuroplasticity is a lifelong process. And in the case of stroke, neuroplasticity is the key to increasing either function or dysfunction in affected limbs.
“Bottom line,” Taub says bluntly, “If you don’t use it, you lose it.”
A therapy with many applications
Deeply encouraged by the results, Taub went to Germany in 2003 to work with several investigators on cortical plasticity, also known as neuroplasticity, the brain’s ability to reorganize itself by forming new neural connections based on new experiences.
In a series of studies on stroke patients, Taub and his colleagues demonstrated that after damage to the nervous system, the brain quietly reorganizes itself. Because of the lack of use of the damaged part of the body, the brain starts pruning back neuronal pathways that are no longer active, while building up new pathways in areas correlating to the expanded use of the unaffected limb.
For example, in a right-handed person affected by a stroke in their right arm and hand, the brain areas related to motor use of the right arm and hand shrink, while the brain areas related to motor function in the left arm and hand (which are now being used) increase in size and activity.
After CI treatment, the process reverses. “After 10 days of treatment we had an enormous increase in the amount of gray matter, the amount of brain tissue involved in the innovation of movements of the affected arm, and that was correlated with the amount of improvement in the arm,” Taub says.
These findings, more than anything else, put CI therapy on the medical map. “Before we published the brain data, there were a lot of people who were skeptical. Forget behavior and movement, forget proof showing range of motion and all the other measures of movement that you can use and that we did use. When we published the brain data, suddenly our findings were real.”
Since first developing the protocol, Taub and his colleagues at the University of Alabama have extended the therapy beyond motor impairment in stroke. CI therapy has also been used successfully to treat stroke patients suffering from aphasia (difficulty with speech), motor and cognitive issues following traumatic brain injury and movement impairments from multiple sclerosis (MS) and cerebral palsy.
In one case, Taub worked with a 55-year-old ophthalmologist who had a stroke that severely impaired his left arm and hand. “He had a moderate deficit, which meant he could only use 10 percent of his arm as compared to before the stroke,” says Taub. “We treated him for two weeks, and his ability to use his arm increased to 50 percent.”
In another case, a young girl of 13 with cerebral palsy came to him because she was suffering from impaired use of her right leg and arm. After two weeks of CI treatment she had experienced a substantial improvement in her motor skills. When she returned at age 17 for three weeks of treatment for aphasia, she went from being able to only speak in sentences of one or two words to achieving a verbal performance that was 47 percent of normal.
As opposed to other rehabilitation therapies used for stroke, CI therapy is most effective when started later. At Taub’s clinic in Birmingham, patients aren’t considered for treatment until about three months after a stroke.
“A lot of people think they need to hurry and start the therapy fast,” says Mary Bowman, an occupational therapist at the University of Alabama Birmingham clinic. “But typically we don’t have them come until about six months after a stroke because the brain is going through so much change.
“After a stroke, the brain is almost in a state of neuro-shock. And later there is so much spontaneous recovery happening, it’s impossible to tell whether it’s simply spontaneous recovery or the therapy that’s working.”
Flying in the face of clinical studies showing that most significant improvement after stroke occurs early on in treatment, Taub says often the most dramatic improvement in patients occurs both well after the stroke and even well after CI treatment. Take, for example, the ophthalmologist he treated.
“I didn’t see or hear from the him for about five years,” says Taub. “Then I got an invitation to his sixtieth birthday party where he celebrated the event by giving a piano concert, playing moderately difficult pieces of Mozart and Brahms for half an hour. Afterwards, I asked my wife, who is a classically trained pianist, if she could tell any difference between his skill with his right or left hand, and she said ‘no.’
“Turns out, after he finished CI therapy, he was so determined to keep up his rehabilitation work that he started piano lessons – an instrument he hadn’t played in 50 years. He kept on rehabilitating himself until he was nearly 100 percent post-stroke.”
Which brings us to yet another major difference between CI therapy and other stroke rehab approaches. In addition to a delayed approach to treatment, CI therapy also focuses on post-treatment follow-up.
Rehabilitation therapists, for the most part, agree that on average only 5 percent of stroke victims actually pursue their own rehabilitation at home once they’ve left the hospital and the clinical rehabilitation setting. Knowing this, Taub developed a post-rehab protocol that he dubbed the Constraint-Induced Therapy Transfer Package.
After the patient goes home following their intensive two-week rehabilitation therapy, their therapist calls them once a week for a month and works with them for an hour over the phone doing their exercises. After the first month, the phone rehab sessions drop to once a month for a year. This simple follow-up procedure, which encourages patients to keep working to improve function, has had a significant impact on these patients’ rehabilitation.
“Basically no matter how long after a stroke, there is still potential for change,” says Bowman. “We’ve had people come in maybe 20 years out. I think one person was even 50 years post-stroke.”
A number of independent clinical studies support Taub’s findings. A 2015 review of the research on CIMT found strong evidence that the technique is highly effective in terms of improving motor function, arm-hand activities and functioning in daily life, both immediately after treatment and at long-term follow-up.1 A meta-analysis of 16 randomized controlled trials also found that CIMT is more effective than traditional rehabilitation therapy for improving upper limb function after acute or sub-acute stroke.2 Beyond the effects of CI therapy on movement, constraint-induced aphasia therapy (CIAT) also improves language performance in stroke patients.3
In addition to its use in stroke patients, CI therapy has shown great promise for people with “hemiparetic” progressive MS, who, similar to some stroke patients, experience significant weakness or difficulty moving on one side of the body.4 A recent clinical trial concluded that CIMT produces “very large improvement in real-world limb use and induced white matter changes” in this population of patients.5
CI therapy also has a positive impact on hand/arm use in children with cerebral palsy6 as well as structural changes in their brains indicative of the growth and proliferation of neurons.7
No matter how soon or how late they arrive for CI therapy, rehabilitation patients undergoing the treatment say it’s intense. “Prior to CI therapy, I had movement but no function with my left hand,” says Madeline N, age 25. “The sessions were very challenging and fatiguing, but I was ready to put in the work.
“Today the functional tasks I can use my hand for don’t look as fluid or as good as they did pre-stroke, but the important thing is I am using it. I can do some things now with my hand, like put my hair up in a high bun and type on the computer. I walk completely independently, even on uneven surfaces, and my balance, weight shifting and sensation have all improved.”
“It’s intense for the patient, and it’s intense for me as a therapist,” agrees Bowman. “Depending on how much movement deficit the person has, the typical delivery of CI therapy now is for about three and a half to four hours every day, five days a week, for two to three weeks.
“That’s a lot of hours a day. But when they leave from being with me each day, they’re not finished. That’s only the beginning of their day. Because they need to go home and use that hand or that arm at home.”
As tough as the work can be, Bowman says there’s nothing like seeing patients improve – especially the ones who have been suffering from chronic paralysis and disuse of a limb for years.
“Sometimes when someone has had a stroke, they have a kind of delayed life after the stroke has happened,” she says. “They might not want to move out of the house like they were originally planning, or they may not start that family or they might have the feeling they shouldn’t go back to work because they don’t know if they could handle it.
“For each kind of patient, we hit it head-on with CI therapy, moving straight into those areas where they are desiring to go, making sure that they have a lot of experiences with the very things they would encounter out in life.
“I’ve had quite a number of people leave and then two months later send pictures back and report on how things have changed in their lives. And that’s very exciting to me as a therapist to see.”
A typical constraint-induced therapy session
“Constraint-induced therapy is all about achieving progress in small steps,” says occupational therapist Mary Bowman.
The daily therapeutic tasks given to patients during the three-and-a-half to four-hour sessions can be very simple, like working on grasping with the index finger and thumb by flipping dominoes over on a table.
Bowman describes what she wants people to do and shows them the movement, giving them encouragement, coaching and feedback of their results.
Whether it’s flipping dominoes over, scooping beans into a bowl or making slices through a mock carrot made of putty, each exercise is performed 10 times, and each pass is timed.
“They’re doing a lot of repetition and it can sound like it’s boring, but actually it’s not,” she says. “Most people get competitive with their own time.
“It’s not that we want patients to rush. But we also want them to push themselves, like ‘Oh, I think I can beat that time and improve my game a little more!’
“When we do that, we tend to hone in on our skills a little bit better and focus better. It helps a person start achieving progress in ways they wouldn’t have if they had not had that attention on that task.
“Typically the best result that patients come up with is about the seventh or eighth trial. Everything comes together as they hone in on their motor skills.”
Stroke indicators
Signs of stroke are the same in women and men:
• A sudden numbness or weakness in the face, arm or leg, especially on one side of the body
• Sudden mental confusion
• Trouble speaking or trouble understanding others’ speech
• Sudden difficulty seeing in one or both eyes
• Sudden difficulty walking, accompanied by dizziness, loss of balance and/or lack of coordination
• Sudden onset of a debilitating headache with no known cause
If you suspect you or someone you know is having a stroke, get immediate medical attention. With strokes, time is of the essence. The most effective emergency medical treatments for stroke have to be administered within the first three hours of the start of symptoms to work optimally. Getting immediate treatment can reduce brain damage and make the difference between more severe debilitation post-stroke and more manageable effects.
Resources
Taub Therapy Clinic:
www.uabmedicine.org/patient-care/treatments/ci-therapy
CI Therapy Research Group:
www.uab.edu/citherapy
| References | |
| 1 | Lancet Neurol, 2015; 14: 224-34 |
| 2 | Neural Regen Res, 2017; 12: 1443-50 |
| 3 | Dement Neuropsychol, 2009; 3: 275-82 |
| 4 | Mult Scler, 2008; 14: 992-4 |
| 5 | Neurorehabil Neural Repair, 2018; 32: 233-41 |
| 6 | Phys Ther, 2009; 89: 1126-41 |
| 7 | Pediatrics, 2013; 131: e1664-9 |